One of the two digital health companies in Boomtown’s current health-tech class, cliexa develops mobile applications that assist users in managing their chronic diseases.
Newswire Official Press Release
cliexa hosted a booth at Rocky Mountain Gastroenterology’s 15th Annual Pow Wow Conference on Saturday, April 9, 2016. Conference was held at Hyatt Regency at Denver Tech Center, Denver, CO.
The GI Pow Wow is designed to provide a gastroenterology & hepatology update to community primary care providers, surgeons, specialists, as well as medical oncologists & radiologists from throughout Colorado and the surrounding region.
Topics included:
- Opioid receptors & implications on treating Intestinal Disorders
- Childhood Obesity & Fatty Liver Treatment
- IBD Treatment update
- HCV update
- Esophageal Cancer Prevention
- Drug-Induced Liver Disease
At a conference with 400 healthcare industry providers, cliexa had a lot of visitors with lot of questions for cliexa-RA which is in Apple store and cliexa-IBD in early May 2016. Lots of physicians showed interest at scientific disease activity scoring which is tracked with both cliexa-RA and cliexa-IBD dynamically.
Our CTO Nathan Blair, CSO Esra Nutku-Bilir and myself have answered questions from different providers. cliexa-IBD for Inflammatory Bowel Disease is using a survey type disease activity scoring whereas cliexa-RA uses DAS28 joint point calculation.
cliexa-IBD has attracted lots of physicians, who are interested in EMR integration with their current systems which is now part of cliexa’s roadmap.
Here are some photos from the exhibition!
Mehmet Kazgan, CEO.
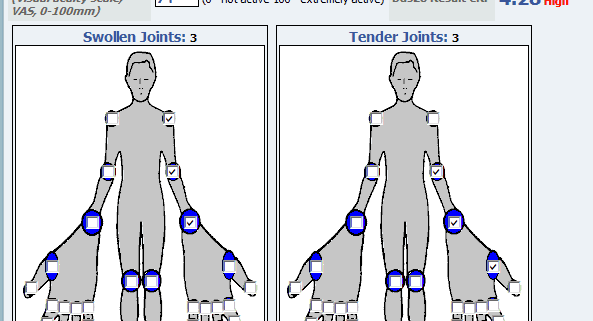
Rheumatoid arthritis (RA) is a chronic inflammatory disease that may impair daily functioning and quality of life due to pain, swelling and stiffness. The disease has an unpredictable course and the main treatment goal is to suppress disease activity in order to prevent joint damage and to improve daily living. Treatment of RA mainly includes disease modifying anti-rheumatoid drugs including biologicals. Both international and national treatment guidelines recommended assessing the disease activity using measures such as DAS28, and doing regular follow up assessments, in which the level of activity dictates the frequency of monitoring. Frequent and regular assessments, however, may not be possible in a clinical practice because of a busy daily schedule.
Healthcare data suggest that Patient Reported Outcomes (PROs) have certain significance for routine clinical care. Following are the questions that we will be discussing here: What is the validity of patient self-report outcomes to guide a treat-to-target strategy in clinical trials and usual clinical care of rheumatoid arthritis? Which measures to use? How could physicians (Rheumatologists) use these measures in a daily clinical practice? Challenges involved, and why DAS28? How could patients contribute to their own treatment?
What is the validity of patient self-report outcomes in clinical practice?
It seems attractive to assess disease activity from patient’s perspective using validated patient reported outcomes (PROs) as an alternative or as an addition to laborious frequent joint assessments. There is evidence concerning PROs in clinical care in documenting low disease activity and remission, including a meta-analysis of studies that document the value of using PROs to implement ‘treat-to-target.’ PROs are well established by groups of experts from the Outcome Measures in Rheumatoid Arthritis Clinical Trials –working group (Felson et al , 1993). Patient global health (pGH) a PRO element is included as a recommendation in the 2010 (ACR-EULAR guideline that proposed tighter definitions for clinical remission in clinical trials, and is now also established for clinical practice (Smolen et al, 2013).
Which measures to use?
Of the 63 currently available RA disease activity measurement tools, the data may be filtered down to 6 recommended measures: the CDAI (clinical disease activity index), DAS28 (ESR and CRP; disease activity score with 28 joint counts) PAS (patient activity scales), PAS-II, RAPID-3 (routine assessment of patient index data), and SDAI (simplified disease activity index). All 6 measures produce a single consistent index and have defined ranges for indicating low, moderate, or high disease activity or clinical remission. Studies demonstrated that by applying these tools systemically in clinical practice, physicians would be able to treat to target and effectively implement the American College of Rheumatology recommendations for the treatment of RA (J. Anderson et al, 2012).
How could physicians (Rheumatologists) use these measures in a daily clinical practice? Challenges involved, and why DAS28?
Realizing the heterogeneity of settings in which health care is delivered to patients with RA in the US, there are selected measures that offer a full range of data collection options. Some are purely patient-reported (PAS, PAS-II, and RAPID3), some need physician assessment (CDAI), and some need physician and laboratory acute phase reactants (SDAI and DAS28). While most of these measures are considered as possible options, purely patient-reported measures may lack formal assessments. Therefore, patient-reported measures must be complemented by a careful joint examination, and do not prevent performance of a formal joint count or any other measure by a treating physician. Meanwhile, DAS28 calculations provide use of a continuous measure with absolute laboratory values and formal physician-assessment in daily clinical practice and clinical trials. Therefore DAS28 has been shown to a useful instrument to monitor disease activity in to titrate treatment with biologicals (Fransen et al, 2005). However, frequent collection of patient data and calculations are challenging in a busy rheumatology practice. There are indications that the DAS28 is not measured frequently enough in standard rheumatology care. Thus, daily, regular, patient self-report joint assessments may provide a useful, cost-effective method to complement physician’s role and implement treat-to-target in patients with RA.
How can you -as a patient- contribute to your own RA- treatment?
Collaborative Network 4 Clinical Excellence (CN4CE) has recently launched an application, cliexa-RA that is based on DAS28 score calculations with erythrocyte sedimentation rate (ESR). Cliexa-RA is available for iOS devices in the Apple store. This, so far unique, application is designed to combine patient-reported measures, with laboratory (ESR values) and provides an essential tool for the physician to review quickly and monitor disease activity, and make a medication titration, even within a busy schedule. This application is designed to collect patient self-reported joint symptom data and medications, daily and frequently, and calculates DAS28 scores automatically by combining the other variables involved in DAS28 scoring system (such as ESR). Physicians act to complement patient’s role by reviewing and confirming the data. Thus, cliexa-RA provides disease activity monitoring tool based on DAS28 scoring system and data on the medication history over a time period. cliexa-RA not only increase awareness, and encourages patients to involve in their own treatment, but also provides an effective support tool for the physicians’ office to increase treatment outcomes and implement treat-to-target in patients with RA.
Esra Nutku-Bilir MD, PhD
Co-Founder, Scientific Advisor
Post Doctoral Award recipient from American Rheumatoid Arthritis Foundation
Current Challenges in Managing Complex Chronic Illnesses
Chronic Care Management, Chronic Disease Activity, Clinician Experience, Digital Health, Evidence-Based Treatment, Mobile Health, Patient Engagement, Patient Experience, Patient Reported Outcomes, Population Health, Preventive Care, Responsive Care, Value-based CareChronic and complex illnesses may represent the minority of the patients, seeking the attention in our healthcare today. However these illnesses consume the majority of the Healthcare expenditure, as they require a specialty care and their complications can be life threatening. While effective therapies are available, without careful monitoring their effectiveness as well as patient or physician adherence to these; devastating complications, loss of productivity and long lasting deformities can happen.
Rheumatoid Arthritis (RA) is one of these chronic illnesses affecting 1. 3 million adults in U. S. population. It is characterized by an autoimmune (patient’s own immune system attacking and leading to an) inflammation in the lining of the joint capsule and the lining of tendons. RA can cause not only crippling chronic joint pain, but can lead to permanent joint damage, and loss of joint function. This illness is a major reason for loss of productivity as well as decreased quality of life in the affected patients. In most cases, the patient is totally dependent on the physician to control their disease activity and have very little participation, other then being compliant to take their medications and keep their appointments. This is what is expected of them and considered to be the standard of care today. When and if their access is somehow interrupted by another illness, or a loss of insurance or changing of their physician; the patient totally loose their ability to monitor the activity of their illness and get lost in the follow up. They can loose their job and insurance due to time loss from work and end up with deformities and complications, only fueling the fire in this vicious cycle.
This is where the Cliexa-RA app can be very helpful to the patient to keep track of their illness, outside the physician clinics. A well proven and accepted Heath care tool, easy enough to be utilized by the patient, will increase their own awareness of the disease activity and seek prompt attention before serious complications develop. In addition, if their physicians obtain their objective and reliable data, before the clinical visit, they can arrange an earlier visit or change the medication comfortably.
The patient utilizing this system will also have a chance to see the effects of the medications which will make them much more compliant with their therapy. So, we anticipate that this application will increase patient compliance and relieve their anxiety of the unknown, before each clinical visit, while allowing their physician to monitor their progress effectively. If this happens, we can hope that this will improve the overall well being and the outcome of the patients utilizing this application.
Bahri Bilir, MD, FACP, FAASLD
Medical Director of Liver Transplantation,
Presbyterian St Luke’s Hospital Denver Colorado